(ii) Concentration of Substrate: (Cone of Soc Rate of E Action up to a limit (E is constant)
(ii) Concentration of Substrate: (Cone of Soc Rate of E Action up to a limit (E is constant)
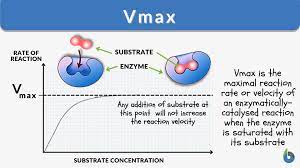
(a) Low Concentration:
• At low concentration of substrate the reaction rate is directly proportional to the substrate available. (b) High Concentration: If the enzyme concentration is kept constant and the amount of substrate is increased, a point is reached when any increase in the substrate does not increase the rate of reaction more, because at high substrate level all the active sites of the enzyme are occupied with substrate.
Further increase in the substrate does not increase the reaction rate, Maximum rate for enzyme at concentrations A and B
A
B
Active altes still available
Active sites almost all occupied. Enzyme is saturated
Saturation of Active side
V max
Rate of reaction
Active site not all occupied
Substrate Concentration
Effect of substrate concentration in the rate of Enzyme-control reaction
Imp. S/Q
Do you know why rate of enzyme action increases at high temperature and decreases at very high
temperature?
Substrate concentration
(iii) Temperature: (Temperature o Rate of E Action upto a limit then rate decreases)
(a) Optimum Temperature:
All enzymes can work at their maximum rate at a specific temperature (7) called as optimum
temperature. For enzyme of human body 37°C is the optimum temperature.
(b) Low Temperature:
At very low temperature (0°C), the activity of the enzyme is minimum.
(c) High Temperature:
Beyond 45°C, usually the activity of enzyme is permanently stopped due to the denaturation (
A) of enzyme. and therefore, chemical reactions are ( ری ایکشن کو شروع کرنے والی توانائی کی کم سے کم مقدار) Heat provides activation energy
• accelerated () at high temperature. So rate of enzyme action increases.
(d) Heat as Kinetic Energy: Heat also supplies kinetic energy to the reacting molecules, causing them to move rapidly, thus the reactants move more quickly (76) and chances of their collision G) with each other are increased.
Tags: Concentration of Substrate
Very good article. I definitely appreciate this website. Stick with it!